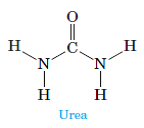
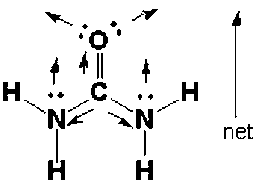
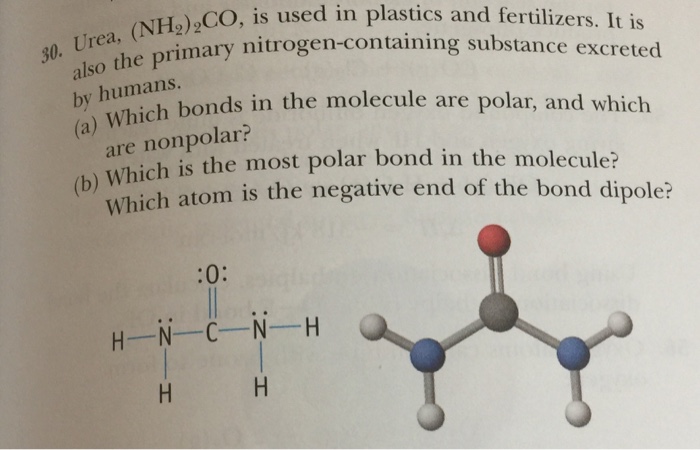
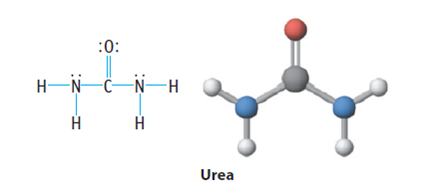
The molecule below is urea, a compound used in plastics and fertilizers. Which is the most polar bond in the molecule? Which atom is the partial negative end of this bond?
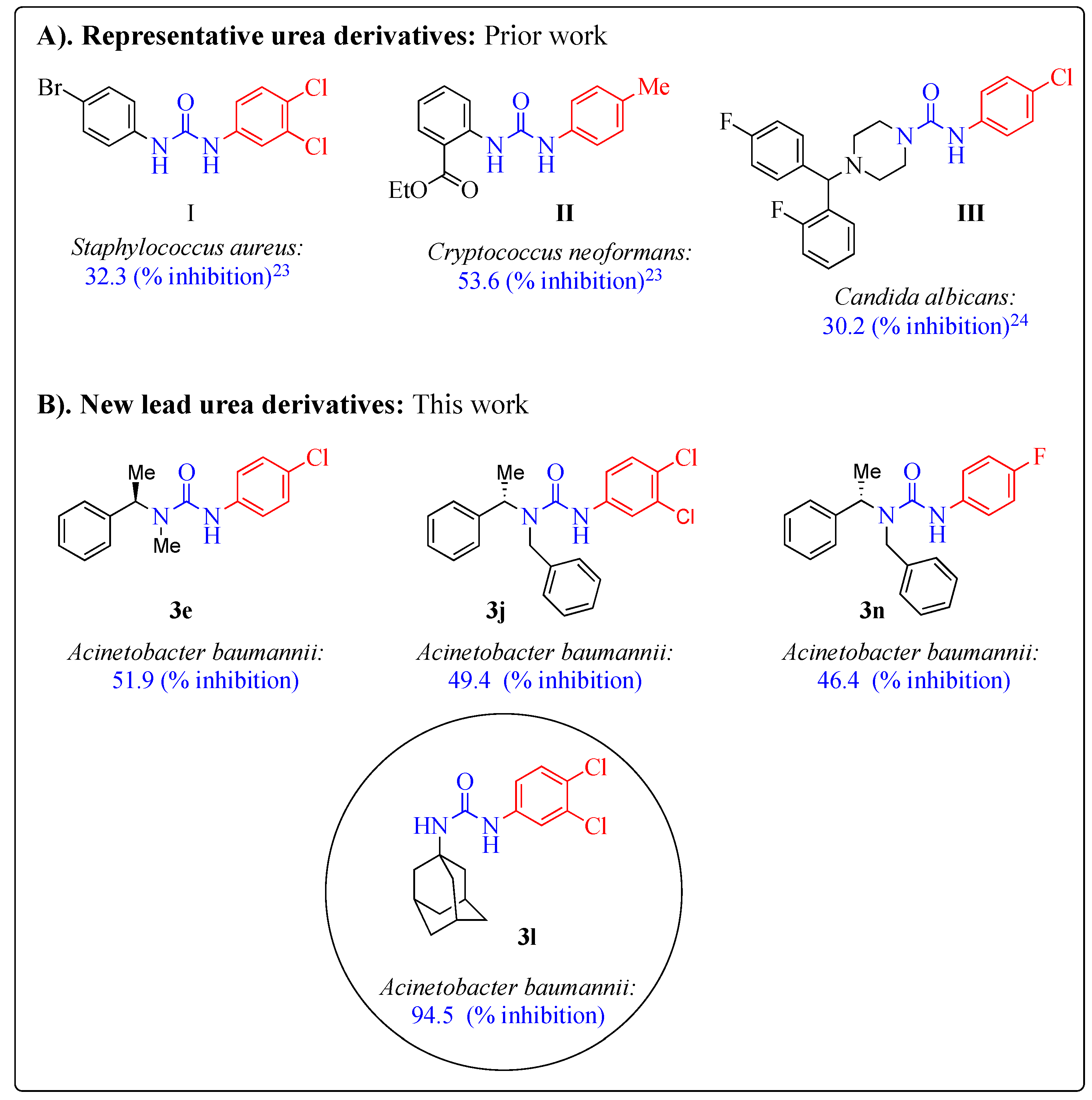
Antibiotics | Free Full-Text | New Urea Derivatives as Potential Antimicrobial Agents: Synthesis, Biological Evaluation, and Molecular Docking Studies

Urea as a Redox-Active Directing Group under Asymmetric Photocatalysis of Iridium-Chiral Borate Ion Pairs | Journal of the American Chemical Society
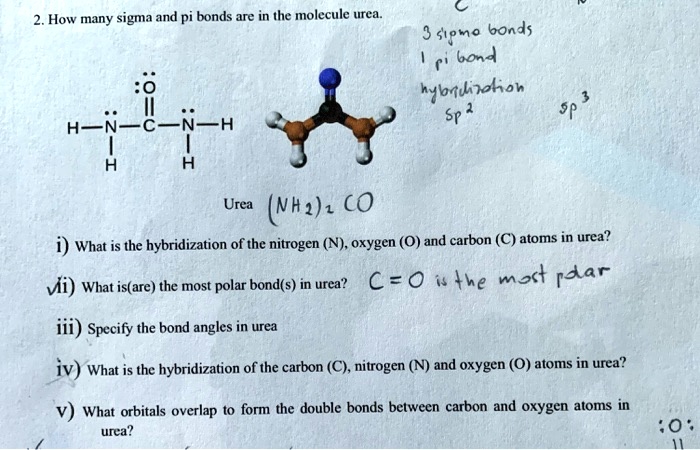
SOLVED: How many sigma and pi bonds are in Ihe molecule urea Spmo bonds 60n4 nybfuioliob Sp H H Urea (Nh2) €0 What is the hybridization of the nitrogen (N) oxygen (0)
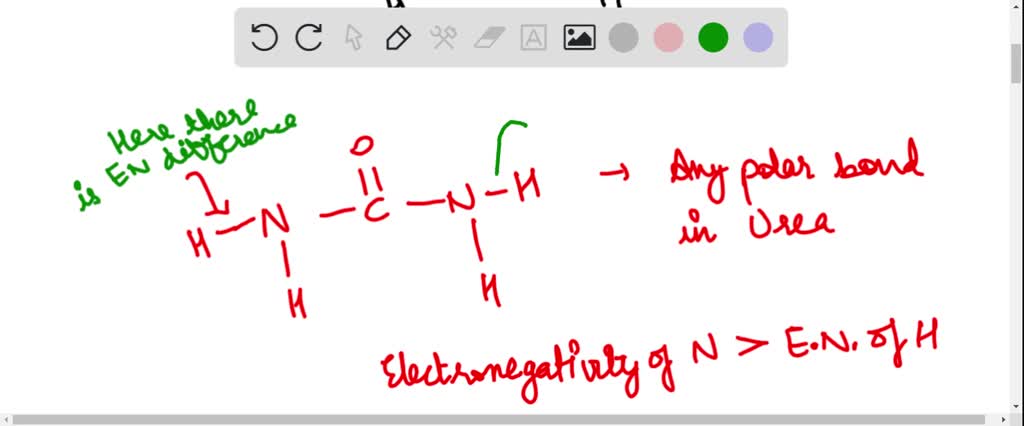
SOLVED: Draw the Lewis structure for urea, H2NCONH2, one of the compounds responsible for the smell of urine. (The central carbon atom is bonded to both nitrogen atoms and to the oxygen


![Polysulfide-assisted urea synthesis from carbon monoxide and ammonia in water [PeerJ] Polysulfide-assisted urea synthesis from carbon monoxide and ammonia in water [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2022/ochem-6/1/fig-4-2x.jpg)