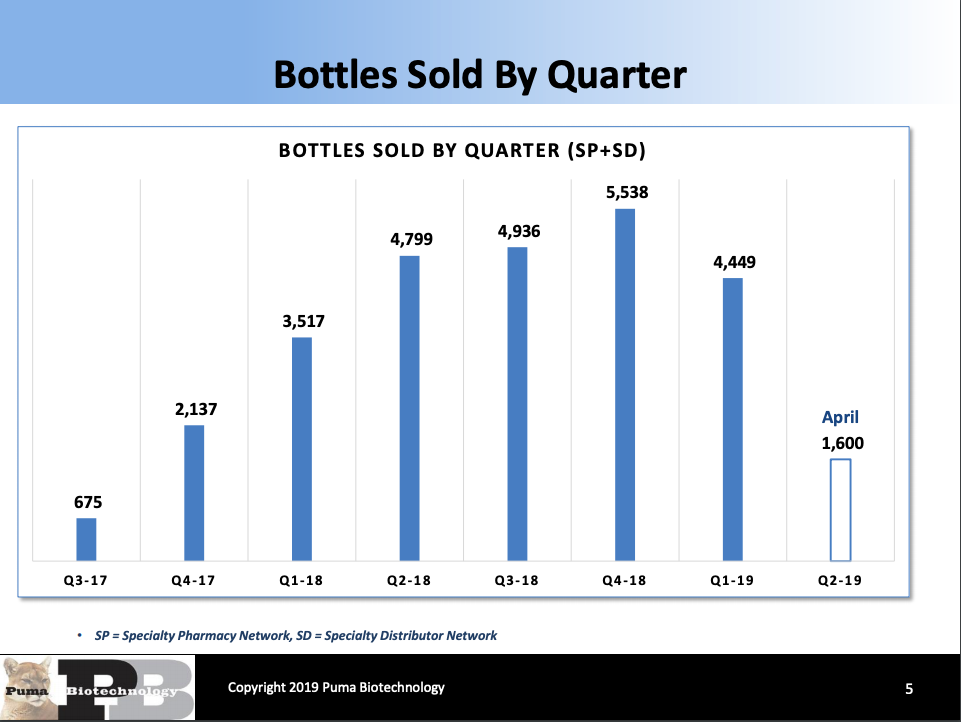
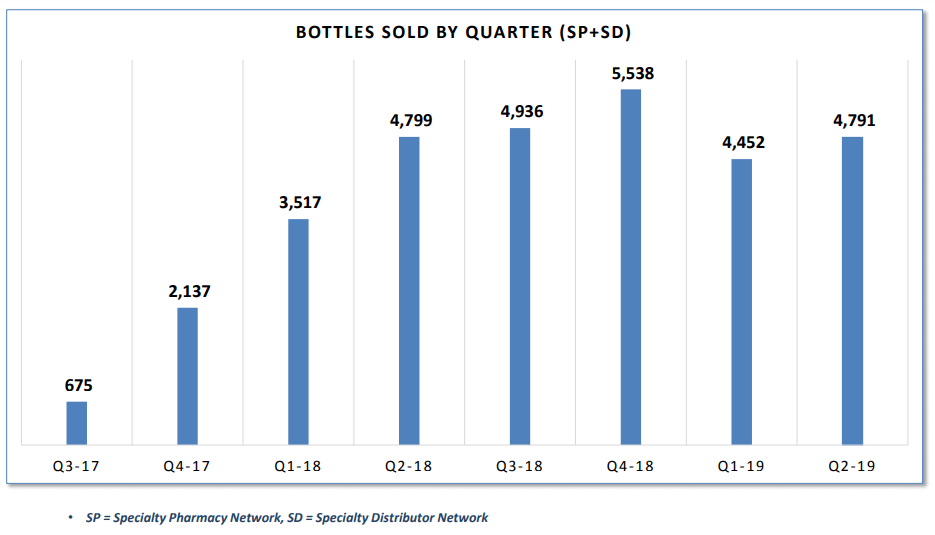
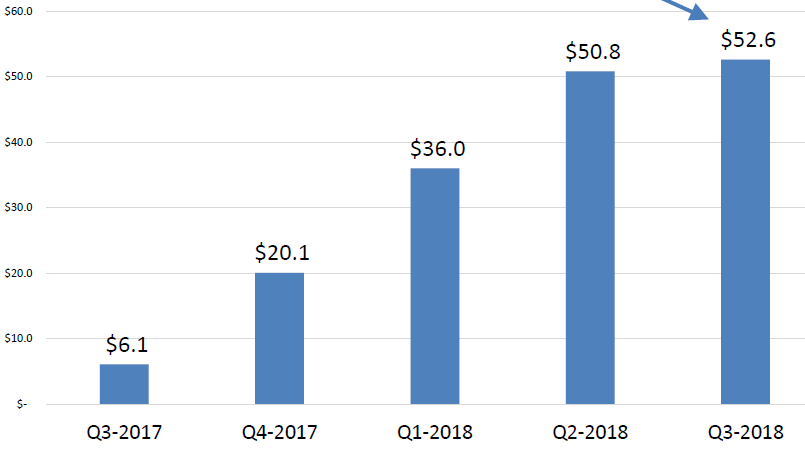Pint-Pharma - Puma Biotechnology and Pint Pharma Enter into Exclusive Licensing Agreement to Commercialize NERLYNX® (neratinib) in Latin America

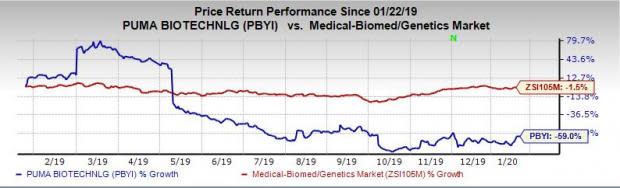
Puma Biotechnology | $PBYI Stock | Shares Climb Up as Company Announced FDA Approval for NERLYNX - Warrior Trading News

These highlights do not include all the information needed to use NERLYNX safely and effectively. See full prescribing information for NERLYNX. NERLYNX ® (neratinib) tablets, for oral use Initial U.S. Approval: 2017

Puma Biotechnology, Knight Therapeutics sign license deal to commercialize NERLYNX in Canada - Pharmaceutical Business review
Puma Biotechnology and Knight Therapeutics Enter into Exclusive License Agreement to Commercialize NERLYNX® (neratinib) in Cana

These highlights do not include all the information needed to use NERLYNX safely and effectively. See full prescribing information for NERLYNX. NERLYNX ® (neratinib) tablets, for oral use Initial U.S. Approval: 2017
FDA Approves Labeling Supplement for Puma Biotechnology's NERLYNX® (neratinib) for the Extended Adjuvant Treatment of HER2-Po