Enhanced hydrogen production via staged catalytic gasification of rice husk using Ca(OH)2 adsorbent and Ce–Ni/γAl2O3 catalyst in a fluidized bed - ScienceDirect

⚗️Complete the following equations (note that the equations are not balanced). Use the activity - Brainly.com

Table 2 from Synthesis of Me Doped Mg(OH)2 Materials for Thermochemical Heat Storage | Semantic Scholar

Figure 2 from Control of uniform nanostructured alpha-Ni(OH)2 with self-assembly sodium dodecyl sulfate templates. | Semantic Scholar
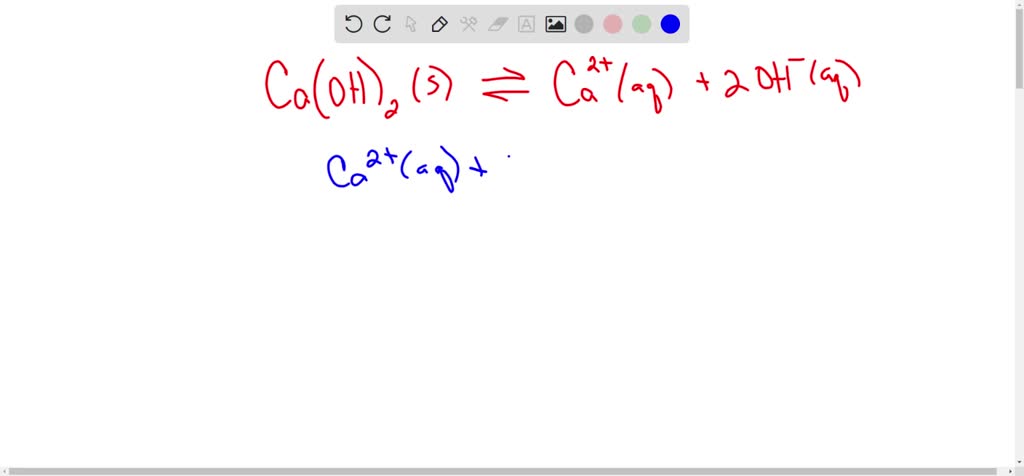
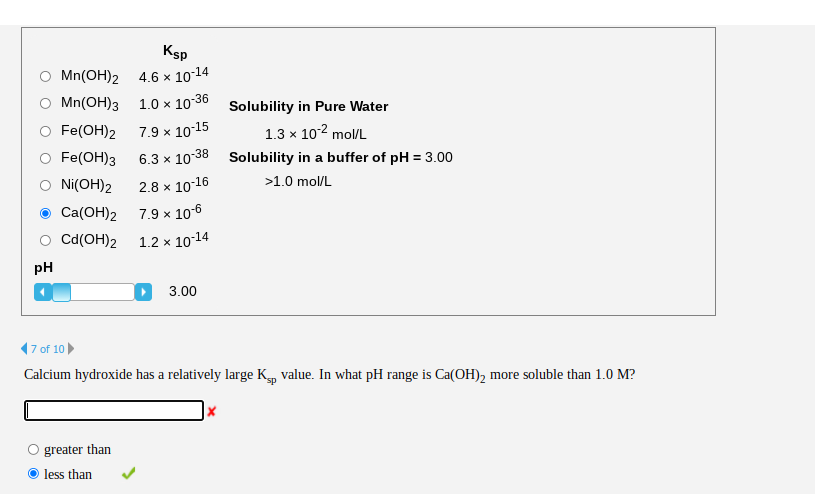
SOLVED: A saturated solution of Ni(OH)2 has the following equilibrium. Ni(OH )2 Ni2+ + 2OH- If NaOH is added to this saturated solution, the amount of Ni (OH)2 in the solution 1) increases as

Figure 3 | Nickel and Zinc Removal from Acid Mine Drainage: Roles of Sludge Surface Area and Neutralising Agents

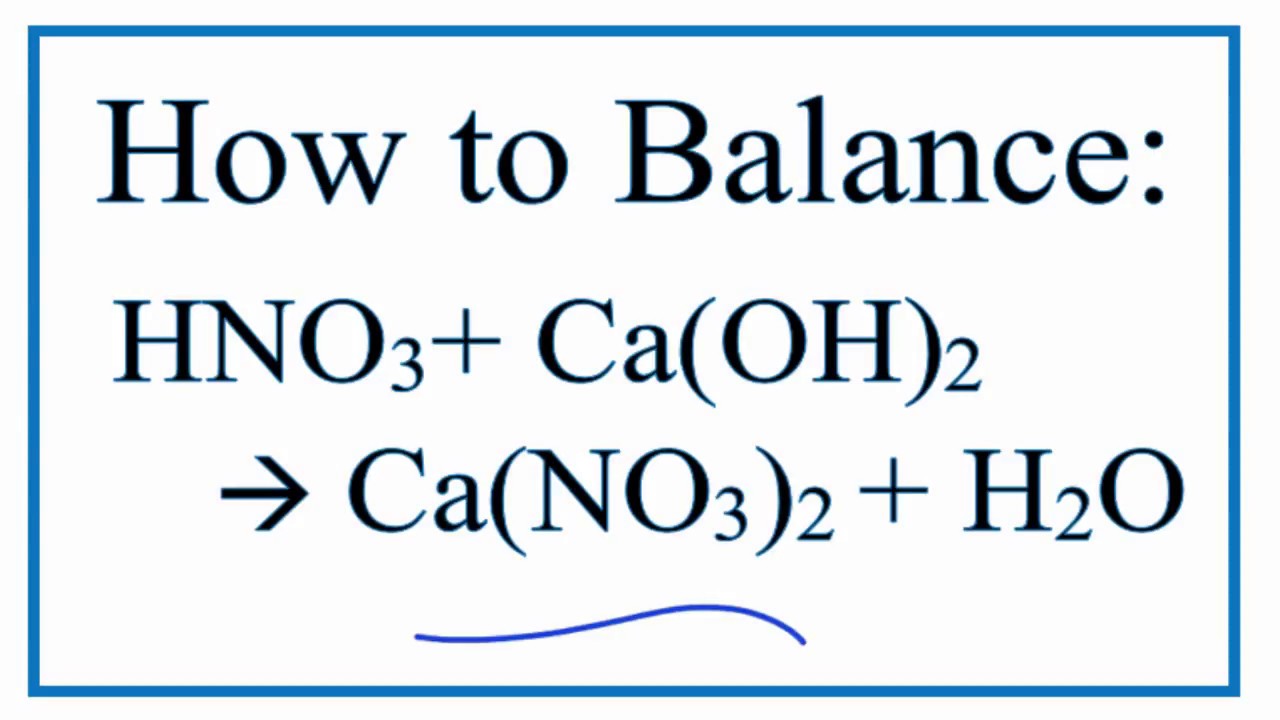
SOLVED: Consider these compounds: A. Fe2S3 B. Caz(PO4)2 C. Ni(CN)2 D. Ca(OH) 2 Complete the following statements by entering the letter(s) corresponding to the correct compound(s). (If more than one compound fits the

Enhancing Hydrogen Evolution Activity in Water Splitting by Tailoring Li+-Ni (OH)2-Pt Interfaces | Science

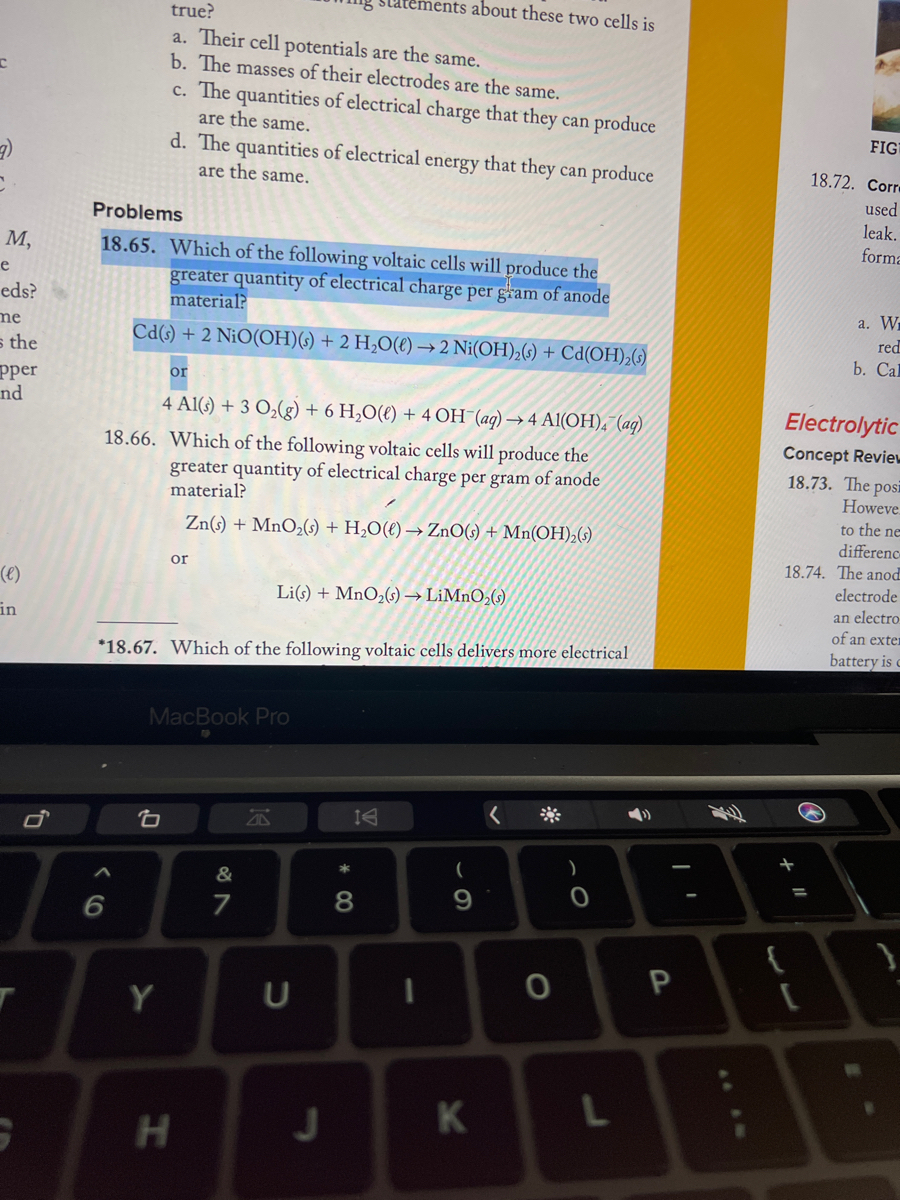
SOLVED: Use the activity series to determine which of these reactions will NOT happa All of these reactions will happen ZnCl2 Ba BaClz Zn Ca Ni(OH)z Ni Ca(OH)z AI(NO3l3 Pb Pb(NO3l2 None