![Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium](https://miro.medium.com/v2/resize:fit:1182/0*DWO_1XXK-IxUiSTt.jpg)
Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
1. Details of Module and its structure Module Detail Subject Name Chemistry Course Name Chemistry 03 (Class XII, Semester 01) Mo
![Explain the type hybridisation, magnetic property and geometry for [ Ni (CN)4 ]^2 - and [ Ni (NH3)4 ]^2 + using VB theory. Explain the type hybridisation, magnetic property and geometry for [ Ni (CN)4 ]^2 - and [ Ni (NH3)4 ]^2 + using VB theory.](https://haygot.s3.amazonaws.com/questions/633645_607318_ans_06ef044c85004ad2aed957fc1fd1f24d.png)
Explain the type hybridisation, magnetic property and geometry for [ Ni (CN)4 ]^2 - and [ Ni (NH3)4 ]^2 + using VB theory.
![Exceptional case, Hybridisation ,shape of an outer orbital complex [ Ni(NH3) 6]2+, Octahedral complex - YouTube Exceptional case, Hybridisation ,shape of an outer orbital complex [ Ni(NH3) 6]2+, Octahedral complex - YouTube](https://i.ytimg.com/vi/R5RDFu1oYUU/maxresdefault.jpg)
Exceptional case, Hybridisation ,shape of an outer orbital complex [ Ni(NH3) 6]2+, Octahedral complex - YouTube
Draw the structures of [Co(NH3)6]^3+, [Ni(CN)4]^2- and [Ni(CO)4]. - Sarthaks eConnect | Largest Online Education Community
Draw the structures of [Co(NH3)6]^3+, [Ni(CN)4]^2- and [Ni(CO)4]. - Sarthaks eConnect | Largest Online Education Community
Ni(NH3)2(NO3)2—A 3-D Network through Bridging Nitrate Units Isolated from the Thermal Decomposition of Nickel Hexammine Dinitr
![Under the valence bond approach explain the shape and magnetic behaviour of [Ni(NH3)6]2+.[Given At. No. of Ni = 28] - Wired Faculty Under the valence bond approach explain the shape and magnetic behaviour of [Ni(NH3)6]2+.[Given At. No. of Ni = 28] - Wired Faculty](https://www.wiredfaculty.com/application/zrc/images/qvar/CHEN12070315.png)

![Answered: [Ni (NH3) 6] Magnetic moment of 2+… | bartleby Answered: [Ni (NH3) 6] Magnetic moment of 2+… | bartleby](https://content.bartleby.com/qna-images/answer/c73807b6-45ed-4420-a3b1-cee7bec70749/203ec893-0265-480e-b058-95e1378e904d/3cda3a30-aca8-11eb-99b8-c16e6153b0d2_IMG_20210504_124206_523~2.jpg)
![why cant [Ni(NH3)6]^2+ have dsp3 hybridization then? : r/CBSE why cant [Ni(NH3)6]^2+ have dsp3 hybridization then? : r/CBSE](https://preview.redd.it/why-cant-ni-nh3-6-2-have-dsp3-hybridization-then-v0-slwfvbv55ria1.png?auto=webp&s=7ae8c48cb10f964fc01fc13df37e5b45cf7e58cf)
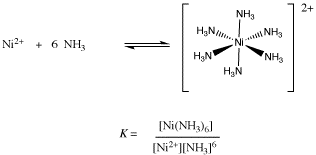
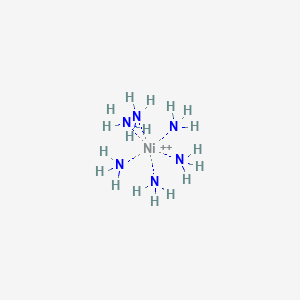

![Answered: [Ni (NH3) 6] C12 Explain the… | bartleby Answered: [Ni (NH3) 6] C12 Explain the… | bartleby](https://content.bartleby.com/qna-images/question/33dc3186-d7b9-4388-8560-215f799e8fe0/013f6e76-4df4-4063-bb5c-5eb17a580f72/z92qakc_thumbnail.png)

![How is [Ni(NH3) 6] 2+ paramagnetic while [Ni(CN) 6] 4- diamagnetic? - Quora How is [Ni(NH3) 6] 2+ paramagnetic while [Ni(CN) 6] 4- diamagnetic? - Quora](https://qph.cf2.quoracdn.net/main-qimg-61bcbbeed69e0a294c52675e216d428f.webp)